The FDA Inspection Management System for Life Sciences
Ready Room is the all-in-one GxP inspection readiness and management platform trusted by biotech, pharmaceutical, medical device, CRO, and CMO quality teams to prepare for and run FDA, EMA, and MHRA inspections.
What is Ready Room?
Ready Room is a cloud-based FDA inspection management system and GxP inspection readiness platform for life sciences companies. Quality assurance teams at biotech and pharmaceutical sponsors, contract research organizations (CROs), contract manufacturing organizations (CMOs), bioanalytical labs, and medical device companies use Ready Room to centralize inspector requests, manage document responses, rehearse a consistent quality story with 200+ GxP storyboard templates, and coordinate front-room and back-room teams in real time during FDA, EMA, MHRA, and other regulatory inspections.
OVERVIEW
Regulatory inspections don’t have to be chaotic.
Ready Room helps life sciences companies streamline GCP, GMP, GVP, and GLP inspection readiness by reducing inspection risk, aligning stakeholders, and bringing organization to a process that is always high stakes. Deliver documents quickly. Answer questions confidently. Tell a consistent quality story in your next inspection.

Why Inspection Readiness Often Fails
Disjointed tools and channels:Instant messaging, shared spreadsheets, and document repositories aren't purpose-built to manage your inspection.
Losing control of requests and documents: No real‑time insight or automation means requests can get lost and teams struggle with collaboration and accountability.
Unaligned teams: When sponsors, CROs, and QA are not in sync, they don't tell the same quality story, adding risk to your inspection.
Inspection readiness and management shouldn't feel chaotic. Ready Room replaces complexity with clarity, so you and your team are always under control.
We will buy out your current inspection management software license
Limited Time Offer
Don’t let your current system hold you back. For a limited time, Ready Room will buy out your existing inspection management software licenses so you can move to a faster, secure, more intuitive inspection readiness platform, without incurring any sunk costs. Don't wait until your current license expires, contact us today to get started. Learn more.

Your Inspection Success Starts Here
• Reduce inspection risk with a proven, centralized system
• Align global teams across sponsors, CROs, consultants, and vendors
• Be inspection-ready anytime, anywhere
• Fast onboarding gets your team up and running in hours
• SOC 2 certified security trusted by leading pharma and biotech companies
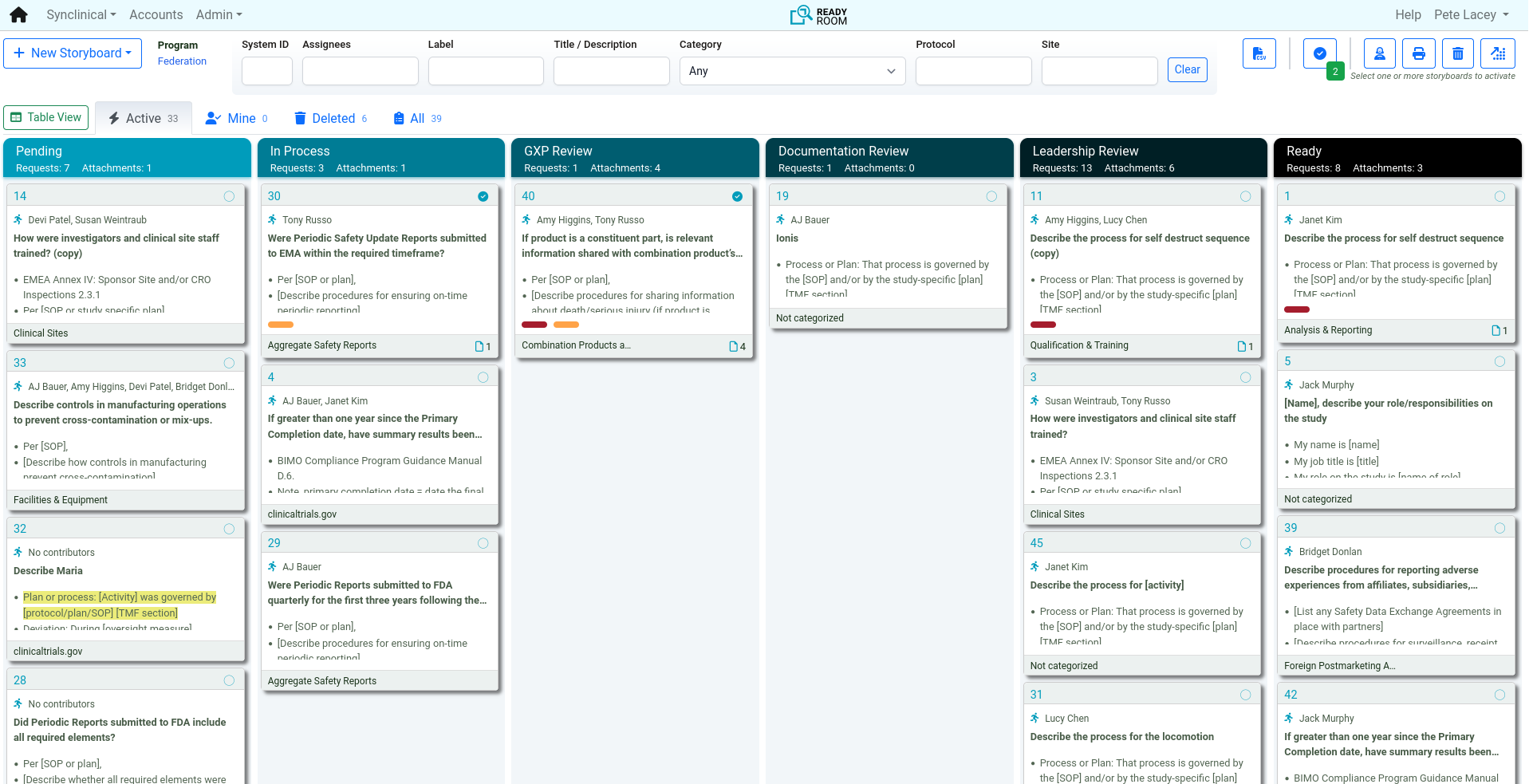
Inspection Readiness: Build and Rehearse Your Quality Story
Inspection readiness is about proactive preparation. With Ready Room, teams use 200+ GxP storyboard templates to align on a consistent quality narrative. A proven workflow and coaching tools ensure everyone is confident and inspection-ready before the first question is asked.
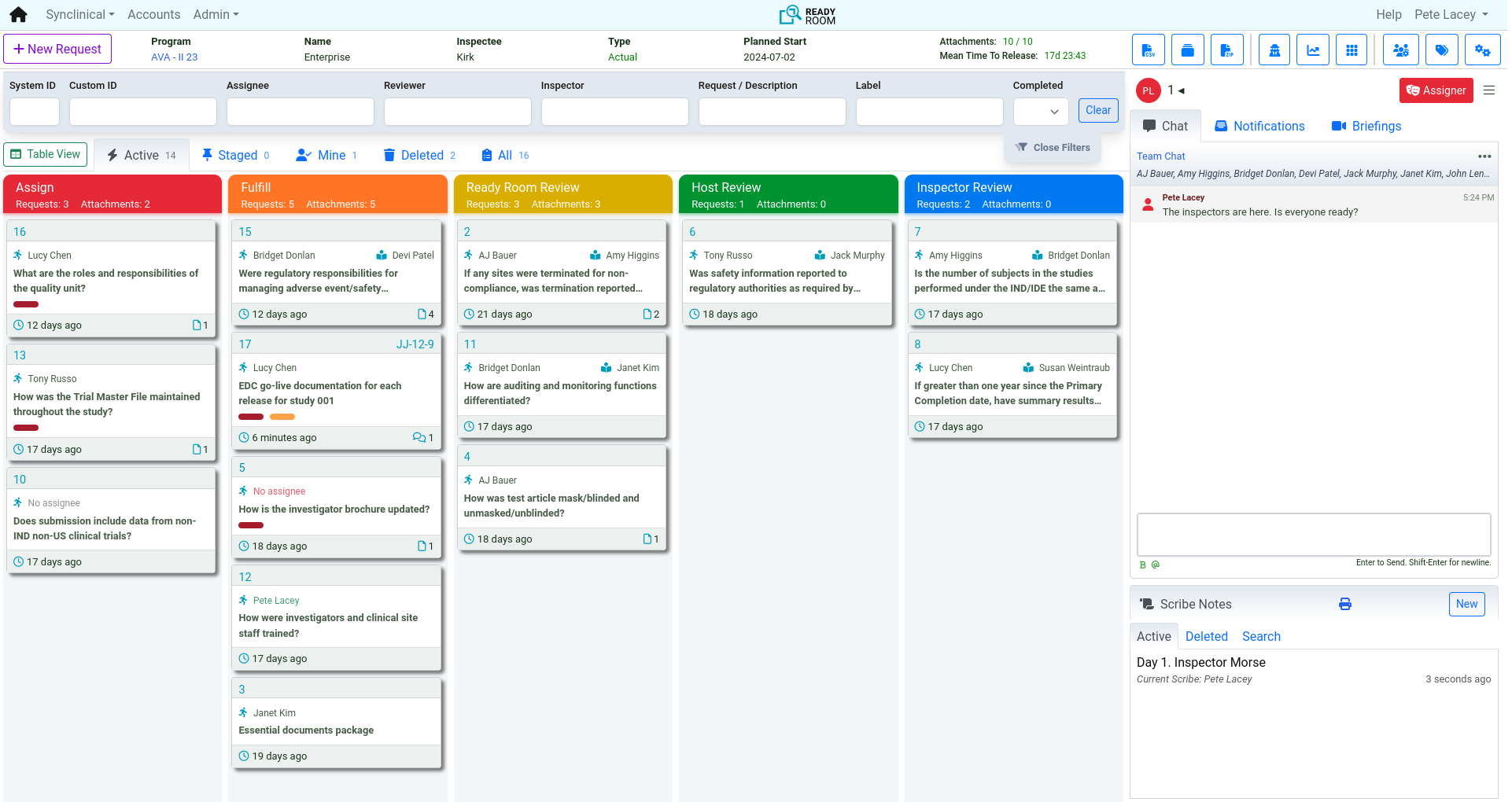
Inspection Management: Execute with Speed and Control
Inspection management begins the weeks before inspectors arrive. Ready Room provides a centralized board to capture, assign, and review every request in real time. With built-in chat, scribe notes, and QC tools, your team stays aligned and in control from start to finish.
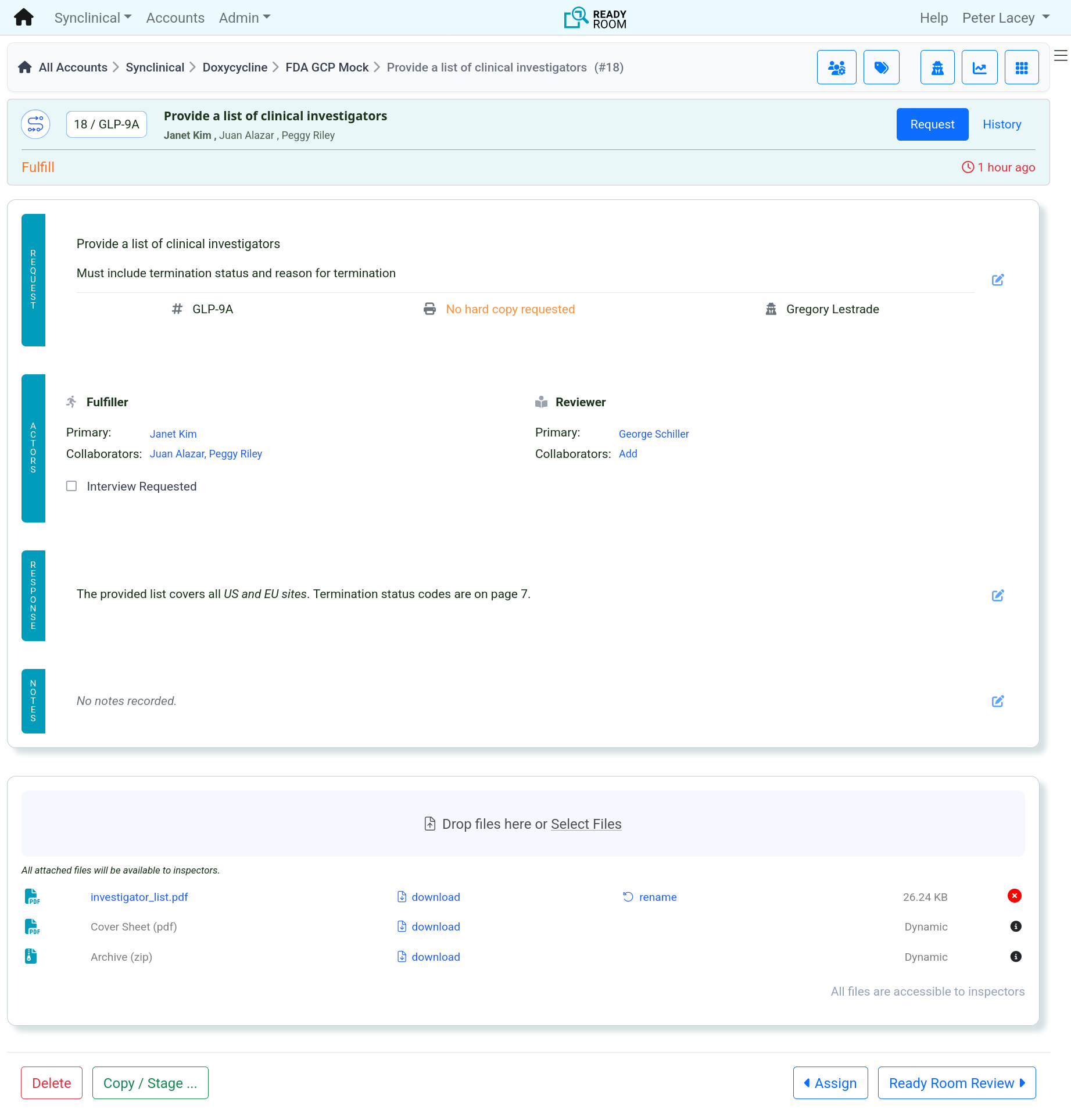
There's more to storyboards than PowerPoint
Inspection Readiness, Reinvented
This 90-second video shows how Ready Room empowers teams to manage every request, storyboard, and response in one powerful platform.

We're Committed to Security
Ready Room is SOC 2 Type II Certified
We understand the critical nature of data security in the highly regulated life sciences industry. Ready Room's SOC 2 Type II certification is a testament to our ongoing dedication to maintaining a secure and trustworthy environment for our customers. We continuously invest in our security infrastructure and processes to meet and exceed industry best practices.

Ready to Get Inspection Ready?
Book a personalized demo or start a two-week free trial for up to eight users.
What our customers are saying
OUR TESTIMONIALS
Latest from the Blog
Trusted GxP Inspection Management for Global Life Sciences Teams
From biotech startups in Boston to pharmaceutical leaders in Basel, Ready Room helps teams stay inspection-ready. Whether you're a sponsor, CRO, CMO, lab, or medical device company, our cloud-based platform simplifies inspection preparation and management across every part of your organization.